
Synaptic Physiology Methods: Experimental Stimuli
During a single experiment, a stereotyped set of stimuli were delivered to recorded cells to characterize the strength, kinetics, and short-term plasticity of synaptic connections. Each stimulus was delivered to cells in turn while recording the response from all other cells. Each stimulus train consisted of eight pulses to induce short-term plasticity (induction) followed by a delay and four pulses to measure recovery from dynamic effects. The frequency of pulses and delay were varied to measure these effects on synaptic connections. There was a 15 second delay between each stimulus to allow the cells to fully recover from stimulation. A separate set of stimuli were delivered to characterize the intrinsic properties of cells.
Stimulus Frequencies
A variety of stimulus frequencies were used, each with a 250 ms delay between the eight induction pulses and four recovery pulses.

Recovery Delays
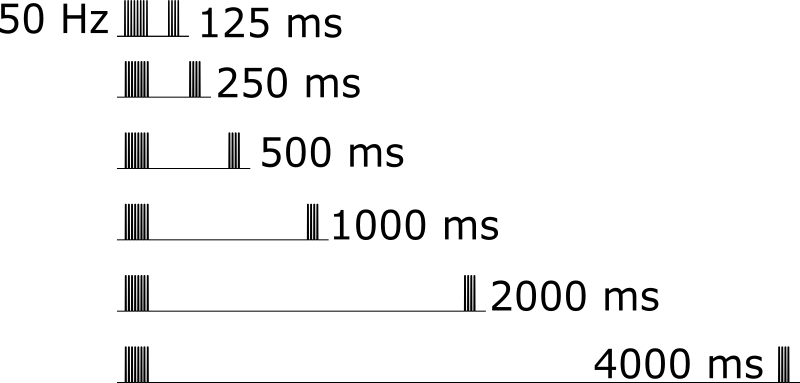
For the 50 Hz stimulus, a range of recovery delays were interposed between the eight induction pulses and four recovery pulses to evaluate the timecourse of recovery from short-term effects.
Mixed Frequency Stimulus
The "mixed frequency" stimulus was composed of eight pulses delivered at 30Hz immediately followed by 30 pulses whose intervals were a random resequencing of 29 exponentially increasing intervals between 5 and 100 ms. The intervals were fixed across sweeps and experiments. This stimulus allowed us to explore a wider range of stimulus frequencies that could be used to inform our model.

Stimulus Trials
Each stimulus, one frequency with one delay or mixed frequency, was repeated at least five times with a 15 second rest period between repeated trials. Spikes were recorded in the presynaptic cell along with postsynaptic responses.

Cell Intrinsic Stimuli
Long, sustained stimuli were delivered to each cell to measure intrinsic properties as well as quantify electrical synapses in current clamp. Intrinsic features were extracted from these stimuli using the IPFX package.
Subthreshold, mostly hyperpolarizing, stimuli were used to measure properties such as input resistance and sag. This stimulus set was initiated with a pulse at -20 pA while keeping the neuron at -70 mV. The voltage response to each current step was measured online and successive current steps were titrated to target response voltages of -68, -72, -75, -80, and -85 mV so as to reliably activate Ih when present.

Suprathreshold, depolarizing stimuli were delivered to measure spiking properties. These stimuli started at rheobase and increased 25 pA for 6 intervals.

A 15-second sinusoidal chirp that increased in frequency from 0.2 to 40 Hz. The amplitude was targeted to evoke a response magnitude that measured ~10 mV from peak to trough.

Your browser is out-of-date!
Update your browser to view this website correctly.